Medical Silicone Tubing: Definition, Uses and Selection Guide

Medical silicone tubing includes extruded silicone conduits used in regulated care and life‑science manufacturing: single‑ and multi‑lumen catheter shafts, peristaltic pump tubing, radiopaque‑marked lines, and reinforced hoses. Procurement and supply teams rely on it for patient‑contact devices and sterile fluid paths where safety, compliance evidence, and repeatable performance matter. In this guide, you’ll learn how the material and cure systems work, which standards govern biocompatibility and sterilization, what construction types exist, how manufacturing and QA controls support capability, and a pragmatic selection workflow you can use to qualify suppliers and specify the right build.
What is Medical Silicone Tubing?
Definition
Medical silicone tubing serves as a flexible, hollow conduit made from silicone polymer. This synthetic material stands out for its exceptional biocompatibility and inertness. Unlike traditional materials such as rubber or plastic, silicone tubing offers a unique combination of properties ideal for medical applications.
Material systems
Two families dominate medical silicone tubing:
-
Liquid Silicone Rubber (LSR): A low‑viscosity, two‑part silicone typically used for molding; certain formulations are extrudable when precise metering is available. LSR grades for medical use are most often platinum‑cured and prized for purity and lot‑to‑lot consistency.
-
High Consistency Rubber (HCR): A gum‑based silicone compound extruded into tubing and cured in continuous ovens. HCR is the mainstay for most tubing and catheter shafts, with medical programs commonly selecting platinum‑cured HCR for low extractables and broad sterilization compatibility.
Cure chemistries
-
Platinum‑cured (addition cure): Produces very low extractables and avoids peroxide byproducts, which is why it’s the default choice for high‑purity fluid paths and many catheter applications. It is broadly compatible with steam, ethylene oxide (EO), and validated radiation cycles when material and function are verified.
-
Peroxide‑cured (free‑radical): Often acceptable for non‑critical applications but may carry higher small‑molecule byproducts, which can raise extractables and limit radiation compatibility. Many programs therefore reserve peroxide cure for non‑contact or auxiliary lines.
For a practical overview of property envelopes used in healthcare tubing programs, see representative data sheets from major suppliers (e.g., NewAge Silcon Medical spec for durometer and tensile ranges) in the section below.
Key properties
Representative ranges (verify on your chosen grade and lot): Shore A hardness typically spans 35–80A, with 50–70A common for flexible lines and peristaltic service; tensile strength often falls near 7–10 MPa with elongation at break of 300–600%+ depending on formulation. Examples appear in the Silcon Medical sheet from NewAge Industries describing 60A tubing and related mechanicals: see the manufacturer’s PDF under “Specifications” here: NewAge Silcon Medical specification sheet.
Compliance basics for medical silicone tubing
Biocompatibility
ISO 10993‑1 frames a risk‑based approach that maps biological endpoints to device contact type and duration (limited, prolonged, long‑term). FDA’s 2023 summary provides a practical, public reference procurement can cite when aligning test expectations with engineering and QA, detailing endpoints such as cytotoxicity, sensitization, irritation, systemic toxicity, and when longer‑term endpoints are considered. See the FDA’s overview in the Biocompatibility Assessment Resource Center: FDA biocompatibility evaluation endpoints (2023).
USP Class VI (USP <88>) is frequently listed on silicone materials, but it is narrower than a device‑level ISO 10993 evaluation and does not equate to device approval. Treat USP Class VI as a material data point; rely on ISO 10993‑1’s risk process for device submissions.
Regulatory references
Medical silicone tubing is typically manufactured under an ISO 13485 quality management system with batch‑level records. For supplier qualification, procurement should expect to review an ISO 13485 certificate, material and finished‑goods Certificates of Analysis (CoA), lot traceability linking raw silicone to finished tubing, and change‑control policies that govern formulation or process modifications in line with FDA’s alignment of 21 CFR 820 to ISO 13485 practices.
Sterilization standards
-
Ethylene oxide (EO): ISO 11135 governs development, validation, and routine control of EO sterilization. The CDC’s EO overview summarizes process elements, and ISO 10993‑7 sets limits for EO residuals and aeration requirements. See: CDC overview of EO sterilization.
-
Radiation (gamma or e‑beam): ISO 11137 defines dose setting and validation (e.g., VDmax 15/25 kGy methods) to achieve an SAL of 10⁻⁶, with maximum dose capped to preserve material function. An open‑access review hosted by the National Library of Medicine explains method selection and verification: ISO 11137 dose‑setting review (NLM, 2023).
-
Steam (moist heat): ISO 17665 specifies validation and routine control of moist‑heat sterilization. An accessible explainer outlines how cycle development and biological indicators fit into validation: ISO 17665 steam sterilization explained by LSO‑Inc.
Types and builds
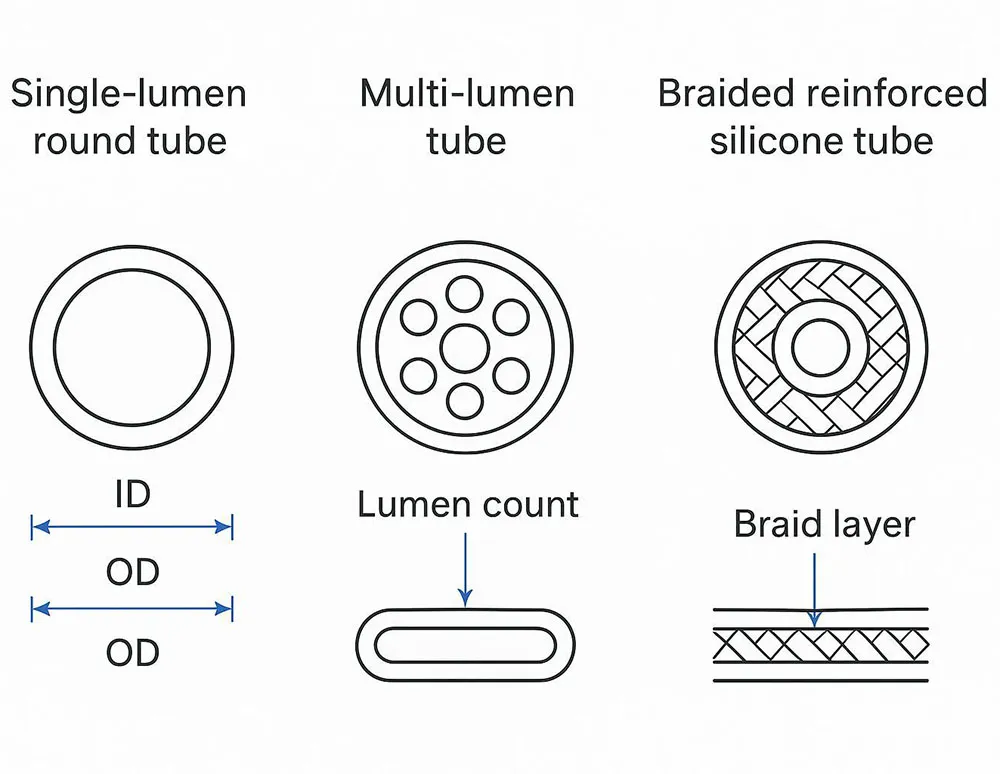
Single vs multi‑lumen
A single‑lumen tube provides one flow path and favors simplicity and cleanability. Multi‑lumen shafts integrate multiple channels in one extrusion to combine functions or segregate fluids; geometries can be tailored (e.g., symmetric or twisted lumens) to manage kink behavior and minimize occlusion in bends.
Reinforced variants
For higher pressures or vacuum lines, a braided or spiral reinforcement layer embedded in the silicone wall improves pressure rating and kink resistance. These constructs are common in anesthesia/breathing circuits and equipment connections where stability and crush recovery are critical.
Radiopaque and pump tubing
Radiopacity can be achieved by compounding radiopaque fillers into silicone or adding discrete radiopaque stripes/markers for X‑ray or fluoroscopic visibility. As a simple real‑world example of a radiopaque marker, see the urinary catheter description that notes a radiopaque strip for visualization under scans in the JESilicone portfolio: radiopaque strip example on a urinary catheter. For peristaltic service, platinum‑cured silicone is widely selected due to purity and elastic recovery; wall thickness, durometer, and pump head settings jointly determine life.
Manufacturing & QA
Cleanroom extrusion
Medical silicone tubing is typically extruded in controlled environments and post‑cured before cutting or spooling. In cleanrooms, processors use non‑contact laser micrometers to monitor OD/ID/wall, manage concentricity and ovality, and apply statistical process control (SPC) with real‑time alarms to maintain capability.
In‑line and SPC checks
Laser systems track dimensional trends and process stability; capable programs often hold OD or wall tolerances on the order of ±0.001 in (≈25 µm) for select sizes and geometries, while complex multilumen builds may target around ±0.002 in across many features—subject to design and equipment capability.
Traceability docs
Procurement should request ISO 13485 certification, raw and finished‑goods CoAs, batch manufacturing records (DHR/DMR elements as applicable), lot traceability from raw polymer to shipped tubing, declared sterilization compatibility or validated cycles, maximum radiation dose (if applicable), and defined change‑control procedures.
JESilicone operates ISO 13485–certified cleanrooms and supports custom silicone extrusion with documentation that aids supplier qualification. JESilicone

Sterilization & effects
Steam and EtO
Under ISO 17665, moist‑heat cycles are validated with biological indicators and process equivalency checks; platinum‑cured silicone commonly tolerates routine autoclave exposure when function is verified post‑cycle. For EO (ISO 11135), validation covers preconditioning, gas exposure, and aeration; because silicone can absorb EO, plans based on ISO 10993‑7 ensure residuals fall within limits without lot‑by‑lot residual testing once the process is validated. The CDC’s EO sterilization page is a concise public reference on process fundamentals and safety controls: CDC overview of EO sterilization.
Gamma/e‑beam
Radiation dose setting per ISO 11137 typically validates minimum doses such as 15 or 25 kGy (VDmax methods) to achieve SAL 10⁻⁶ and establishes a maximum dose to protect material properties. Peer‑reviewed overviews note that silicone elastomers may exhibit modest hardness/modulus increases at validated doses due to crosslinking, while higher or cumulative doses can promote chain scission that reduces elongation and tensile strength; always confirm function after sterilization and over intended life: ISO 11137 dose‑setting review (NLM, 2023).
Property changes
|
Modality |
Typical standard |
Common effects on silicone (directional) |
|
Steam/autoclave |
ISO 17665 |
Generally compatible; prolonged dry heat can harden silicone (↑ durometer, ↓ elongation). |
|
Ethylene oxide |
ISO 11135 (+ ISO 10993‑7 residuals) |
Compatible; may require extended aeration due to absorbency, especially in long lumens. |
|
Gamma/e‑beam |
ISO 11137 |
Validate min/max dose; small ↑ in hardness/modulus possible; excessive/cumulative dose may reduce elasticity. |
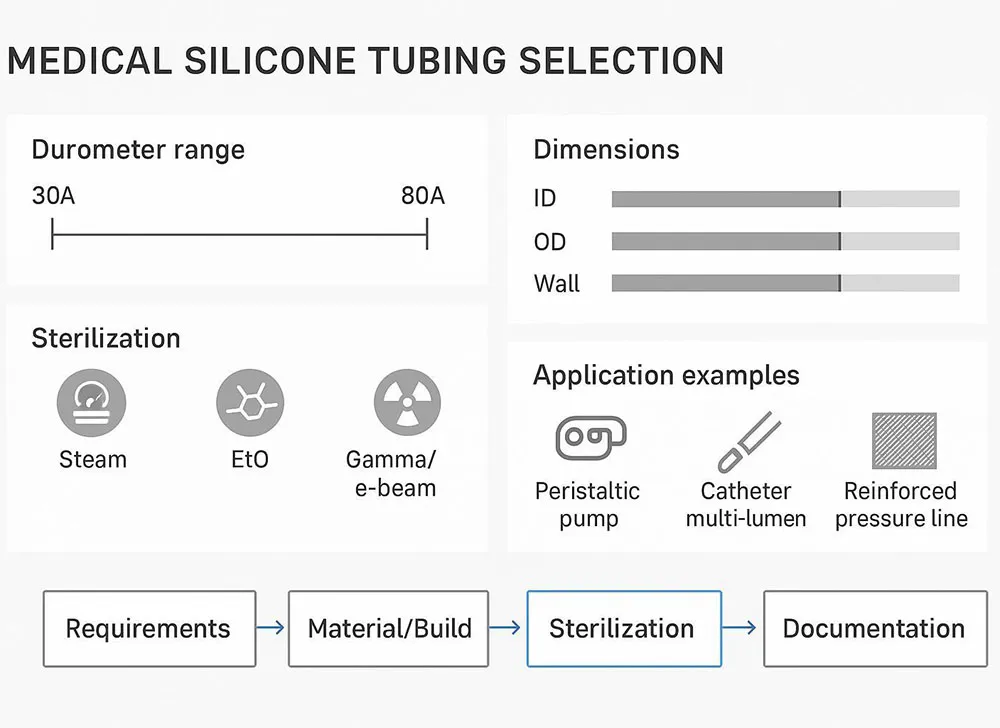
Selection Guide - How to Choose The Right Medical Silicone Tubing
Sizing & mechanics
Define ID, OD, and wall first. For many medical programs, precision extrusion can hold OD/wall tolerances near ±0.001 in (≈25 µm) on select simple geometries; multilumen parts often target around ±0.002 in depending on lumen count and spacing. Choose durometer to balance flexibility and kink/pressure resistance: 50A–60A for flexible transfer lines and pump heads; 70A–80A where clamp load and pressure stability dominate. Use braided or spiral reinforcement for high‑pressure or vacuum duty.
Compliance checklist
For RFQs and supplier audits, request at minimum:
-
Material evidence: USP Class VI summary and ISO 10993 biological evaluation report/summary appropriate to device contact and duration.
-
QMS: ISO 13485 certificate; documented change‑control policy.
-
Batch documentation: CoAs (raw and finished), DHR/DMR elements as applicable, and lot traceability mapping.
-
Sterilization: Declared compatibility and any validated cycles; EO residuals plan per ISO 10993‑7; radiation maximum dose if applicable.
For a concise view of sizing ranges and hardness options, see JESilicone’s capabilities overview for custom medical and pharmaceutical tubing: custom medical silicone tube capabilities.
Application fit
-
Peristaltic pumps: Larger bore at lower speed generally extends tube life by reducing occlusion fatigue; confirm with your pump OEM’s guidance and functional tests. See the manufacturer’s guidance here: Watson‑Marlow tubing guidance.
-
Radiopacity: For imaging visibility, specify a radiopaque compound or add a radiopaque stripe/marker and confirm visibility under your imaging energy and wall thickness conditions. A simple marker example appears in JESilicone’s catheter reference linked earlier.
-
High‑pressure lines: Select reinforced silicone (braid or spiral) and higher durometer; validate burst, vacuum collapse, and clamp retention in your assembly conditions.
Silicone vs. PVC/TPE
Advantages
Silicone’s advantages include broad sterilization compatibility (steam, EO, and validated radiation), thermal stability, elastic recovery with good kink resistance, and a long track record in bioprocess and patient‑contact applications. These traits make medical silicone tubing a strong candidate for sterile transfer lines and peristaltic pump circuits where purity and resilience are priorities.
Trade‑offs
Compared with PVC or many TPEs, silicone typically costs more, may have higher gas permeability, and soft grades can kink without reinforcement. By contrast, some TPEs offer weldability and lower cost for single‑use assemblies, while PVC (with appropriate plasticizer controls) remains common in cost‑sensitive, non‑implant lines.
When to choose which
Choose medical silicone tubing for high‑purity, high‑temperature, multi‑sterilization, and peristaltic pump lines—especially where low extractables and elastic recovery are important. Consider TPE where tubing must be weldable/sealable in single‑use manifolds and cost is a primary driver. Consider PVC for non‑implant, low‑heat applications with validated plasticizer and leachables controls.
Conclusion
To specify and buy confidently, align tubing design with patient risk, process needs, and applicable standards. Start with contact type and duration (ISO 10993‑1), select a platinum‑cured silicone where purity and sterilization breadth are required, and match geometry/durometer/reinforcement to mechanics and pump duty. Then validate sterilization (ISO 11135/11137/17665 as applicable), confirm post‑sterilization function, and lock documentation: ISO 13485 evidence, CoAs, lot traceability, and change‑control. Next steps: qualify at least two suppliers, run functional and sterilization verification on your worst‑case builds, and file a clean document set for audits.