Top 10 Silicone Vaginal Dilator Manufacturers 2026

Introduction
Who this guide is for: sourcing managers, medtech teams, and pelvic health brands
If you're sourcing silicone vaginal dilators products, the hard part usually isn’t finding “a supplier.” The hard part is qualifying a supplier that can support regulated-market requirements—documentation, process control, traceability, and scale—without surprises after validation.
This guide is written for:
-
sourcing and procurement managers building supplier shortlists
-
medtech engineering and QA/RA teams responsible for supplier qualification
-
pelvic health brands that need an OEM/ODM partner for design, tooling, and scalable production
Note: This article is not medical advice. It focuses on supplier qualification and compliance expectations for device manufacturing programs.
How we evaluated silicone vaginal dilator manufacturers and what’s new in 2026
Our evaluation is built around verification-first signals: quality system maturity, risk-based documentation, and evidence you can request during onboarding (not marketing claims).
What’s new in 2026 is that the compliance bar is effectively higher for global programs—even when the product design is stable—because quality system expectations and traceability maturity are increasing in both the U.S. and EU.
How to use this guide to build a compliant, scalable supplier shortlist
Use this list as a starting point, then run a consistent vetting workflow:
-
Identify 3–5 candidates that match your product profile (OTC vs prescription, sterile vs non-sterile, reusable vs single-use).
-
Request evidence in the same format from each vendor (certificates, test reports, traceability approach).
-
Use a matrix to compare “can they show it?” rather than “can they say it?”
Methodology & selection criteria
Scope and data sources
This roundup focuses on manufacturers and brands associated with silicone vaginal dilators in global markets.
We used:
-
publicly available information (company websites, distributor listings, regulatory guidance)
-
standards and regulator documentation (FDA, EU MDR guidance, ISO biocompatibility frameworks)
-
a compliance-led OEM/ODM lens: the manufacturer’s ability to support documentation, change control, and scalable production
Verification levels and evidence
Because manufacturer claims vary in detail, we grouped evidence into three verification levels you can request:
-
Level 1 — Basic identity & quality posture: ISO 13485 certificate (scope matters), quality manual summary, complaint handling overview.
-
Level 2 — Program readiness: incoming inspection controls, process validation approach, device history record (DHR) samples (redacted), change control procedure, supplier management procedure.
-
Level 3 — Regulated-market depth: biocompatibility rationale for the finished device configuration, sterilization validation package (if applicable), UDI readiness and label control, post-market surveillance interface (for branded programs).
OEM/ODM and quality metrics
For silicone vaginal dilators, the most practical metrics tend to be process- and documentation-oriented:
-
Materials control: resin traceability, COAs, colorant/additive control, contamination risk controls
-
Manufacturing environment: clean area/cleanroom practices when required by the program (verify class and scope)
-
Process capability: molding consistency, surface finish control, lot segregation, rework rules
-
Quality system maturity: ISO 13485 alignment, CAPA effectiveness, supplier controls, training records
2026 regulatory and market shifts
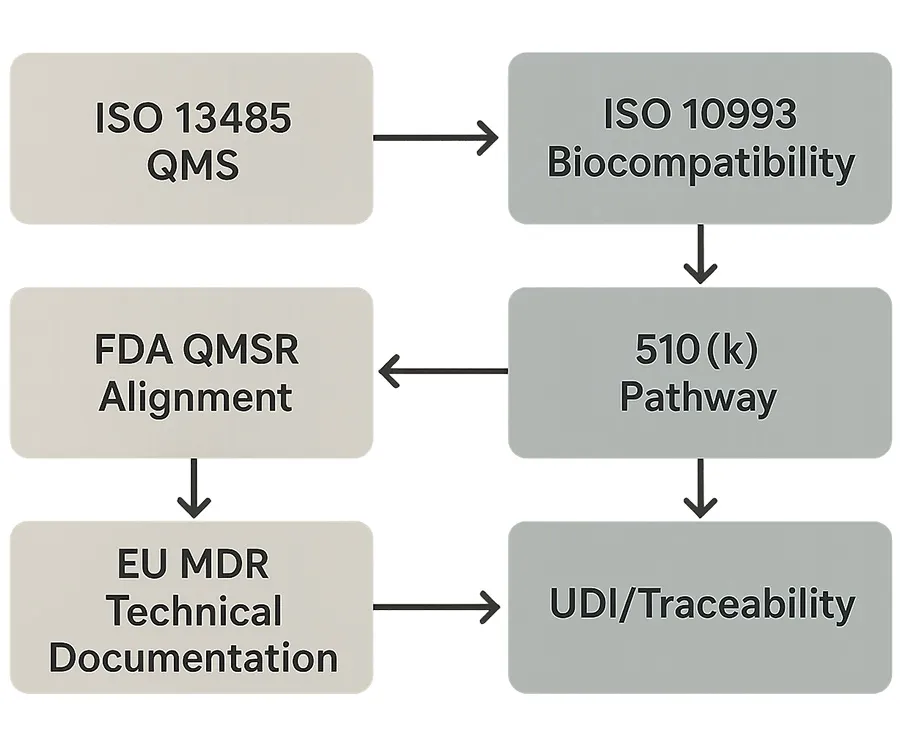
FDA Class II, QMSR alignment, and 510(k)
Many pelvic health devices (including dilator systems, depending on intended use/claims and classification) are typically managed under U.S. medical device rules that expect a compliant quality system and appropriate premarket pathway.
A major compliance milestone is the FDA’s Quality Management System Regulation (QMSR), which becomes enforceable in 2026. The FDA describes QMSR as aligning U.S. quality system requirements by incorporating ISO 13485:2016 by reference in 21 CFR Part 820 (with FDA-specific clarifications). See the FDA’s Quality Management System Regulation (QMSR) overview and the Federal Register final rule (2024) for the canonical details.
For sourcing teams, the practical implication is simple: expect more pressure to show evidence, not just state that a system exists—especially around risk-based supplier controls, complaint handling, CAPA, and change control.
EU MDR IIa, CE marking, and UDI/traceability
In the EU, MDR requirements continue to drive deeper technical documentation, post-market surveillance expectations, and traceability.
On traceability specifically, the MDR UDI framework is supported by MDCG guidance documents. Two high-value references for supplier vetting are the MDCG 2022-7 Q&A on the UDI system and the MDCG 2021-19 guidance on integrating UDI within the QMS.
For buyers, this translates into concrete questions:
-
Can the supplier explain their UDI-DI / UDI-PI approach and what triggers a new UDI-DI?
-
Can they show label control and revision controls that tie back to change control?
-
Can they demonstrate lot/serial traceability across raw materials, WIP, and finished goods?
Biocompatibility, sterilization, and sustainability
Biocompatibility is not “a material claim.” It’s a finished-device evaluation tied to contact type, duration, processing, and (when relevant) sterilization.
The FDA’s guidance on ISO 10993-1 biological evaluation (2023) describes a risk-based approach that considers the device’s patient-contacting nature and exposure. For silicone programs, it’s common to combine test evidence with chemical characterization and toxicological risk assessment—depending on device design and risk profile.
Sterilization validation and packaging validation, when required, should be treated as program-specific. If a vendor claims “sterile-ready,” confirm the exact modality (e.g., EtO, gamma, e-beam), validation scope, and packaging system assumptions.
Top 10 Silicone Vaginal Dilator Manufacturers All Over The World
Leaders: JESilicone; Soul Source; CooperSurgical (Milex)
JESilicone
JESilicone is positioned as an OEM/ODM silicone manufacturer with experience in medical-grade silicone production. JESilicone is also the original manufacturer for several well-known silicone dilator brands, such as Bien, Intimate Rose, Alexander Institute, etc. For regulated programs, the most relevant capabilities to verify include ISO 13485 scope, clean production controls for the specific product line, and documentation depth (change control, lot traceability, inspection records).
What typically matters for a dilator program:
-
ability to support custom design and tooling while maintaining controlled revisions
-
material traceability (resin COA/lot linkage) and controlled surface finish requirements
-
consistent lot segregation and quality records suitable for audits
Soul Source
Soul Source is widely associated with vaginal dilator products and is often shortlisted by pelvic health brands because it is specialized in the category and has mature product-market presence.
For sourcing, treat this as a brand/manufacturer relationship question:
-
confirm manufacturing location(s) and quality system scope
-
request evidence for material controls and finished-device biocompatibility rationale
-
validate packaging/label controls if the program needs UDI readiness
CooperSurgical (Milex)
CooperSurgical (Milex) is a recognized medical device supplier in women’s health categories. For procurement teams, the advantage is often familiarity with regulated procurement requirements and established quality system expectations.
Verification focus:
-
documented quality system and complaint/post-market interfaces (as applicable)
-
consistent product identifiers, labeling controls, and traceability support
-
clear change notification processes for long lifecycle programs
Challengers: Intimate Rose; Freudenberg Medical; Bein Aaustralia
Intimate Rose
Intimate Rose is known for consumer-facing pelvic health products and has publicly emphasized medical-grade materials for dilator sets.
If you’re sourcing for a regulated, scalable program, confirm:
-
whether the product you’re evaluating is a cleared medical device vs a wellness product (this can vary by SKU and market)
-
what objective test evidence exists for the finished device configuration
-
how the supply chain supports consistent lot-level traceability
Freudenberg Medical
Freudenberg Medical is a contract manufacturer known in medical technology supply chains and may be relevant for silicone component manufacturing and assembly work under ISO 13485-aligned systems.
Fit considerations:
-
best suited when your program needs industrialized process validation, supplier management, and scale discipline
-
confirm whether they manufacture complete dilator products or supply components/subassemblies in your architecture
Bein Aaustralia
Bein Australia is associated with pelvic health product distribution and sourcing in certain markets.
For supplier qualification, clarify early:
-
whether they are the legal manufacturer, a distributor, or an OEM sourcing partner
-
which entity owns the technical documentation and quality system responsibilities
Specialists: BioMoi; Pelvi; Vaginismus.com; Joydivision
BioMoi
BioMoi is typically positioned as a specialized brand in pelvic health products.
Verification focus:
-
clarity on manufacturing partner(s) and evidence ownership
-
finished-device documentation availability (not just material claims)
-
labeling and traceability readiness for target markets
Pelvi
Pelvi is often referenced in the context of pelvic floor therapy products.
Fit considerations:
Vaginismus.com
Vaginismus.com is commonly recognized for educational resources and product offerings in vaginismus support.
Sourcing lens:
-
confirm who manufactures the physical devices
-
request evidence of consistent manufacturing controls, lot traceability, and change control practices
Joydivision
Joydivision is associated with intimate health products in global markets.
Verification focus:
-
CE/UDI readiness for EU commercialization (where applicable)
-
documentation and traceability across lots, packaging, and labeling
-
biocompatibility rationale for the final, as-sold configuration
Comparison matrix & sourcing workflow
Quality and compliance factors
A practical comparison matrix for dilator manufacturers should make “evidence” the unit of comparison. Example factors:
-
ISO 13485 certificate scope and surveillance status
-
complaint handling / CAPA overview (high-level) and change control procedure
-
incoming inspection and material traceability (resin lots → finished lots)
-
cleanliness controls appropriate to intended use and packaging (verify cleanroom claims)
-
biocompatibility evaluation approach aligned to ISO 10993-1 (device-contact category and duration)
Manufacturing capabilities and documentation
Document-ready manufacturing capability tends to matter more than “capacity” statements.
Evaluate whether the supplier can provide (at minimum):
-
controlled drawings and revision history (including mold/tooling revisions)
-
process flow and critical-to-quality (CTQ) controls for surface finish and geometry
-
inspection plans and acceptance criteria tied to your device specs
-
lot traceability, nonconformance handling, and rework rules
-
packaging and labeling controls that support UDI requirements when applicable
Commercial terms and risk management
Commercial terms should support compliance, not undermine it. A risk-aware workflow typically includes:
-
NDA + quality agreement (defines responsibilities for documentation, change notifications, audits)
-
sample plan with agreed acceptance criteria and lot traceability
-
supplier audit (remote first, then on-site for finalists)
-
change control terms (notification windows, validation support for changes)
-
business continuity (secondary tooling strategy, safety stock agreements where justified)
To make this concrete without turning it into promotion: an OEM/ODM partner such as JESilicone can be evaluated alongside others on the same checklist—OEM/ODM responsiveness, medical-grade silicone processing know-how, ISO 13485 scope, clean production controls, and the ability to supply audit-ready records. What matters is the evidence package you can obtain and the supplier’s willingness to operate under a quality agreement.
Pro Tip: Ask every finalist for the same three artifacts upfront: (1) ISO 13485 certificate + scope, (2) a redacted change-control procedure, and (3) an example lot traceability record that links raw material lots to finished goods lots.
Conclusion
Start with three verifiable candidates and request certificates and audit access
Start with 3 candidates you can verify quickly. If a vendor can’t provide basic certificates, scope clarity, and audit access (even remotely), it’s usually a sign the onboarding effort will be high.
Confirm biocompatibility, sterilization validation, and UDI readiness before PO
Before you issue a purchase order for a regulated-market program, confirm:
-
biocompatibility approach appropriate to contact type and duration
-
sterilization validation package (when applicable) and packaging assumptions
-
UDI/traceability readiness, including labeling controls and lot/serial data integrity
Use the workflow to de-risk scale-up and revisit suppliers as regulations evolve
Treat supplier qualification as an ongoing system: validate once, monitor continuously, and revisit suppliers as regulations, materials, and market expectations change.