Silicone Foley Catheters vs Latex Catheters: Which One Is Better for Patient Care?
Introduction
Urinary catheterization is a routine intervention in modern healthcare—used to drain the bladder when patients cannot void, to support perioperative care when clinically indicated, and to monitor urine output in selected critical-care situations. It is also a well-established risk factor for complications, especially catheter-associated urinary tract infection (CAUTI). For clinical teams, best practice focuses on appropriate indications, aseptic insertion, a closed drainage system, and prompt removal when no longer needed.
For Procurement, QA, and Engineering teams, the practical question is how to choose devices that align with these clinical goals while meeting supplier, documentation, and reliability requirements. Material choice is one of the few catheter variables that procurement can influence directly.
The silicone catheter vs latex catheter decision is often framed as a simple pros/cons debate. In reality, it’s a set of tradeoffs that depend on dwell time, latex-sensitivity policies, obstruction/encrustation history (“blockers” vs “non-blockers”), and design differences such as coated latex versus 100% silicone.
This guide provides a criteria-based Foley catheter materials comparison. You’ll get a grounded view of how silicone and latex Foley catheters differ, where each typically fits, and how to build a defensible selection approach—especially when stakeholders are asking for the best catheter for long-term use.
It’s designed to help you write clear specs, reduce exceptions, and standardize evaluation across sites.
Clinical note: This content supports sourcing and education. Patient-specific catheter selection and catheter management should follow facility policy and qualified clinical judgment.
What Is a Foley Catheter?
Definition and purpose
A Foley catheter is an indwelling urinary catheter designed for continuous bladder drainage. It remains in place because a balloon near the distal tip is inflated after insertion.
The Foley catheter’s purpose is straightforward: provide a controlled drainage path from bladder to collection system when spontaneous voiding is not possible or when drainage/measurement is required.
Common medical applications
Common settings include:
-
Acute urinary retention or bladder outlet obstruction
-
Selected perioperative indications
-
Selected ICU/critical-care monitoring needs
-
Some urology, OB/GYN, and postoperative pathways per institutional protocols
For procurement, these use cases matter because they imply different expected dwell times, different patient populations (including latex sensitivity risk), and different complication profiles.
Key components (balloon, drainage tube, etc.)
A typical Foley catheter includes:
-
Tip: inserted through the urethra into the bladder
-
Drainage tube/lumen: carries urine to the bag
-
Balloon: inflatable retention component
-
Inflation lumen/valve: used to inflate/deflate the balloon
-
Connector: interface to drainage bag/tubing
Material choice can influence stiffness, lumen geometry at a given French size, long-term surface behavior, and compatibility with coatings.
Types of Foley catheters based on materials
When discussing materials, you’ll typically see:
-
100% silicone Foley catheters
-
latex Foley catheters (natural rubber latex)
-
coated latex catheters (e.g., silicone-coated or hydrogel-coated latex)
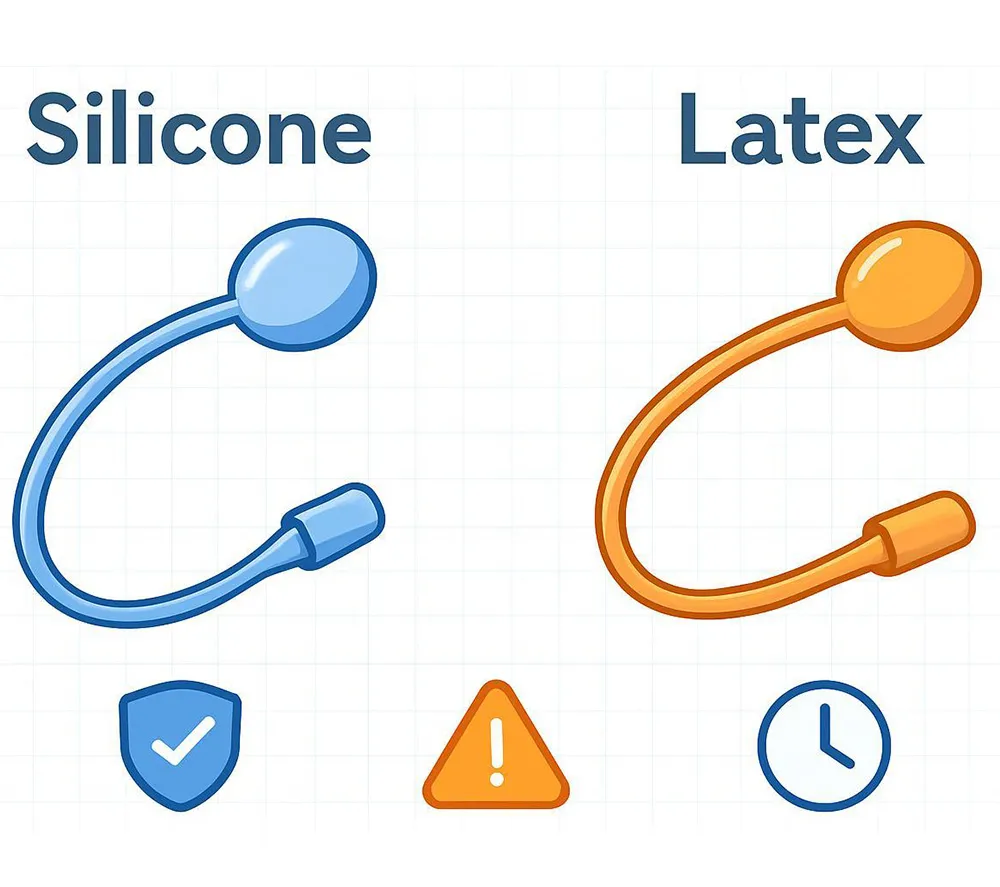
This distinction is essential for risk management: silicone-coated latex is still latex-based, and policies for latex-allergic patients often treat it differently from fully latex-free (100% silicone) options.
Understanding Silicone Foley Catheters
What is medical-grade silicone?
Medical-grade silicone is a synthetic elastomer used in many healthcare devices. In catheter applications, silicone is valued for latex-free composition and material stability in clinical environments.
From a QA perspective, “medical-grade” should be verified through the supplier’s documentation package (material disclosure, biocompatibility test summaries as applicable, IFU/contraindications, and quality-system evidence), not assumed from marketing language.
Key features of silicone Foley catheters
In a sourcing context, commonly cited silicone catheter benefits include:
-
Latex-free material (supports latex-allergy avoidance programs)
-
Suitability for longer dwell times in many protocols
-
Potential advantage in frequent-obstruction patients
One procurement-relevant point from infection-control guidance: the CDC notes that silicone might be preferable to other catheter materials to reduce the risk of encrustation in long-term catheterized patients who have frequent obstruction (Category II) in the CDC’s CAUTI guideline (2019).
Common applications
Silicone Foley catheters are commonly selected when:
-
A patient has known/suspected latex sensitivity or allergy
-
Long-term catheterization is expected
-
A unit wants to standardize on latex-free options
-
Clinicians are evaluating options for patients with frequent obstruction/encrustation
For example, JESilicone’s 100% silicone product family includes a 2-way 100% silicone Foley balloon catheter and a 3-way 100% silicone Foley catheter, which can be relevant when facilities need multiple configurations.
<div data-type="node-video" data-provider="youtube" data-url="https://www.youtube.com/watch?v=MkUG5-mhxko" data-embed-url="https://www.youtube.com/embed/MkUG5-mhxko"></div>
Understanding Latex Catheters
What is latex and how it is used in catheters
Latex (natural rubber latex) is an elastomer used in many medical devices. In Foley catheters, latex is commonly associated with high flexibility and broad availability, and it remains widely used—particularly for short-term catheterization where protocols aim for early removal.
The key limitation is that latex is a known allergen, and mucosal contact increases the likelihood and severity of reactions in sensitized individuals.
Coated vs uncoated latex catheters
Latex Foley catheters may be:
Coatings can improve insertion comfort and reduce friction-related irritation. However, coated latex catheters remain latex-based and may still be excluded by allergy policies.
Typical applications
Latex catheters are often considered when:
-
Catheterization is planned to be short-term
-
The patient has no latex sensitivity history
-
Cost constraints and clinician preference favor latex
In many facilities, latex (often coated) becomes the default short-term option, while 100% silicone is reserved for latex avoidance or longer-term scenarios.
Silicone vs Latex Catheters: Key Differences
This chapter focuses on silicone vs latex catheter differences that procurement/QA teams can translate into purchasing criteria.
|
Decision dimension |
100% silicone Foley catheter |
latex Foley catheter (uncoated/coated) |
|
Material composition |
Latex-free silicone elastomer |
Latex base material; coating may be silicone/hydrogel |
|
Patient comfort |
Often acceptable; depends on size, technique, lubrication, securement |
Highly flexible; coated latex often lowers friction at insertion |
|
Allergy risk |
Supports latex-allergy avoidance |
Latex allergy risk may require exclusion in sensitive patients |
|
Durability & longevity |
Often selected for longer dwell times |
Common in short-term use; long-term use may be limited by policy/complications |
|
Infection risk (practical) |
Management practices dominate; surface may influence colonization/biofilm |
Same; management dominates; coatings may change friction and surface behavior |
|
Cost |
Often higher unit cost; may reduce changes in some scenarios |
Often lower unit cost; coated variants can be mid-range |
Material Composition
A clear Foley catheter materials comparison starts with the base construction:
-
If you need a latex-free device category, specify 100% silicone.
-
If you allow latex, specify whether you accept uncoated latex, silicone-coated latex, or hydrogel-coated latex.
Procurement documents should require explicit material disclosure. “Silicone catheter” is sometimes used loosely in the market to mean “silicone-coated latex,” which is not equivalent from an allergy-risk standpoint.
Patient Comfort
Comfort is multifactorial (French size selection, lubrication, insertion technique, securement, drainage setup, and patient anatomy).
Material influences comfort mainly through:
Coated latex can perform well for insertion comfort in short-term use. Silicone can be well tolerated, especially when size selection and securement are done well. If comfort is a key performance requirement, a structured clinician feedback pilot (rather than assumptions) is usually the most reliable procurement approach.
Allergy Risk
Allergy risk can be a hard constraint.
A safety advisory from the Pennsylvania Patient Safety Authority identified latex indwelling urinary catheters as a common source of inadvertent exposure in patients with documented latex allergy (see Latex: A Lingering and Lurking Safety Risk). For patient-facing education, MedlinePlus latex allergy guidance (2024) notes that mucous membranes can react when latex touches them.
Practical procurement implication:
Durability and Longevity
When teams ask about the best catheter for long-term use, they are usually trying to reduce:
-
Unplanned catheter changes
-
Obstruction episodes related to encrustation
-
Patient discomfort or complications associated with prolonged dwell
CDC guidance provides a narrow but useful decision point: in long-term catheterized patients with frequent obstruction, silicone might be preferable to reduce encrustation risk (Category II) per the CDC CAUTI guideline PDF.
For procurement, this supports a reasonable policy option such as:
Infection Risk
CAUTI risk is strongly driven by catheter duration and catheter-care practices. That’s why guidance emphasizes appropriate indication and early removal, summarized in the CDC CAUTI Summary of Recommendations.
Material and surface can influence colonization and biofilm formation in controlled settings. For example, a peer-reviewed comparison study reported differences in bacterial colonization and biofilm formation across silicone, latex, and silicone-coated latex catheters (see Differences in Bacterial Colonization and Biofilm Formation… (2016)).
For decision-makers, the safest interpretation is:
Cost Comparison
Cost decisions should be framed as total cost of ownership rather than unit price alone.
A practical TCO lens for a silicone catheter vs latex catheter evaluation:
-
Replacement cadence: planned change intervals and unplanned changes due to blockage
-
Policy costs: managing latex-allergy risk (including inadvertent exposure events)
-
Standardization: fewer SKUs and clearer clinical pathways can reduce training burden and errors
-
Supply risk: continuity, lead times, and documentation responsiveness
Pro Tip: Ask suppliers to support your evaluation with a “documentation pack” plus samples for a controlled pilot—then measure unplanned changes and user feedback rather than relying on assumptions.
Advantages & Disadvantages: Silicone vs Latex Catheters
Advantages of Silicone Foley Catheters
Key advantages often cited in a Foley catheter materials comparison include:
-
Latex-free composition, which supports allergy-risk policies
-
Long-term obstruction/encrustation relevance in frequent-obstruction patients, consistent with CDC’s note on silicone in the CAUTI guideline
-
Material stability for longer dwell-time scenarios
Operationally, silicone can simplify purchasing policies in environments where latex avoidance is a first-order requirement.
Advantages of Latex Catheters
Latex catheters can be appropriate when:
-
Use is short-term and the patient has no latex sensitivity
-
Clinicians value maximum flexibility
-
Coated latex options are preferred for insertion smoothness
Latex often remains attractive for short planned durations because the benefits of longer-term materials are not always realized.
Disadvantages of Silicone Foley Catheters
Common tradeoffs include:
-
Different handling feel (often described as less “soft” than latex depending on design)
-
Higher unit cost in many markets
-
Not a universal CAUTI solution: catheter management and duration remain dominant drivers
Disadvantages of Latex Catheters
Key limitations include:
-
Allergy risk and the operational complexity of avoiding inadvertent exposure
-
Long-term use challenges in facilities with frequent obstruction/encrustation concerns and in populations where latex avoidance is required
-
Coating is not equivalence: coated latex remains latex-based and may not fit latex-allergy policies
Clinical Use Cases: When to Choose Silicone vs Latex
This section summarizes practical “fit” scenarios while keeping clinician decision-making central.
When silicone is commonly selected
When latex (often coated) is commonly selected
-
Planned short-term catheterization
-
No latex sensitivity history
-
Clinical teams prioritize flexibility and the facility’s risk review supports latex use
Evidence quality across long-term indwelling catheter comparisons can be heterogeneous and patient-population dependent, which is why many organizations treat material choice as policy plus clinician discretion rather than a one-size-fits-all rule (see Types of indwelling urinary catheters for long-term bladder drainage).
How to Choose the Right Catheter for Your Needs
For Procurement/QA/Engineering teams, a defensible selection is specific, documented, and aligned with clinical practice.
A practical selection checklist
-
Define intended dwell time bands
-
Set the latex policy boundary
-
Specify material disclosure requirements
-
Align to CAUTI prevention practices
-
Ensure your internal policy aligns to CDC prevention priorities (indication, asepsis, closed drainage, early removal).
-
Add an obstruction/encrustation decision rule
-
Pilot and measure
Supplier evaluation criteria (beyond the catheter itself)
If you’re evaluating suppliers (not just materials), include:
For example, JESilicone’s medical silicone portfolio includes silicone catheter and balloon products (see Medical silicone catheters and balloons) and component capabilities such as custom medical silicone tubing, which can matter for OEM integration.
FAQs
What’s the simplest way to frame silicone catheter vs latex catheter selection?
Start with three filters: (1) latex policy/allergy risk, (2) expected dwell time, and (3) obstruction/encrustation history. Then pilot and measure clinician feedback and unplanned changes.
What are the most important silicone catheter benefits in long-term scenarios?
Two are most relevant for policy: latex-free composition (allergy risk management) and the CDC’s note that silicone may be preferable to reduce encrustation in long-term catheterized patients with frequent obstruction.
Are coated latex catheters equivalent to 100% silicone?
No. Coated latex catheters can improve insertion smoothness and reduce friction, but they remain latex-based and may not fit latex-allergy avoidance policies.
Does material choice replace CAUTI prevention practices?
No. CAUTI risk is heavily driven by catheter duration and catheter-care practices. Material can be evaluated as one lever within a broader prevention bundle.
What documents should procurement teams require from suppliers?
At minimum: IFU/contraindications, material disclosure, relevant test summaries where applicable, and QMS/traceability evidence. For supplier maturity, ISO 13485 alignment is a common expectation.
Conclusion
A high-quality silicone catheter vs latex catheter decision is not a slogan—it’s a sourcing policy tied to latex risk, dwell time, obstruction history, and your CAUTI-prevention pathway. For long-term patients with frequent obstruction, CDC guidance notes silicone may be preferable to reduce encrustation. For short-term use where latex isn’t contraindicated, latex (often coated) can remain a practical option.
JESilicone supports OEM/ODM medical silicone products—including silicone Foley catheters and medical silicone tubing—with 20+ years of industry experience, ISO 13485-certified quality systems, and a 100,000-class cleanroom for controlled production. If you are looking for silicone foley catheters, please feel free to contact us for sample and quotation.