How to choose an injection molding manufacturer in 2026

Introduction
If you’re a procurement or supply chain manager, this guide will help you confidently compare and select an injection molding partner, balancing total cost, quality, and schedule. You’ll learn a practical framework to evaluate suppliers side by side, quantify total cost of ownership (TCO), and plan realistic validation timelines. We’ll keep engineers and compliance stakeholders in the loop with what to ask for—without turning your RFQ into a standards textbook.
Use this framework to line up vendors on common criteria, require comparable DFM and validation deliverables, and model the trade‑offs among tooling, unit price, and cycle time. Think of it as your playbook for how to choose an injection molding manufacturer while avoiding apples-to-oranges quotes.
What’s new in 2026? Three shifts shape decisions: (1) FDA’s Quality Management System Regulation (QMSR) aligns U.S. medical device QMS expectations to ISO 13485 starting February 2026, raising the bar for documentation and audits; see the Federal Register final rule and the FDA’s QMSR page for scope and timing. (2) Digital DFM and simulation reports (e.g., Fill/Pack/Warpage and cooling analysis) have become standard buyer expectations. (3) On/nearshoring is now a board-level lever for resilience, while sustainability documentation (e.g., ISCC PLUS mass balance, Scope 1–3 accounting) increasingly rides alongside quality packs.
Selection framework: how to choose an injection molding manufacturer
Define success metrics
Success starts with measurable targets: TCO across the tooling–unit price–cycle time triangle; on-time delivery to gates (T0/T1, FAI/PPAP, IQ/OQ/PQ); dimensional capability indices and cosmetic standards; capacity headroom and scalability; supplier resilience (regional options, dual-sourcing readiness); and sustainability documentation maturity. Make these explicit in your RFQ and request evidence for each.
When this service fits
Injection molding is the preferred process for repeatable plastic parts at scale when you need tight tolerances, consistent cosmetics, and stable unit economics. Aluminum tools often win for prototypes and bridge runs thanks to speed and lower upfront cost. Steel molds suit sustained production, abrasive or high-temperature resins, and multi-million-cycle lifetimes. Hybrids—steel inserts in aluminum frames—can speed learning while protecting wear points.
Comparison rubric
Line suppliers up on a common rubric: capabilities (machines, materials, processes); certifications and regulatory alignment; quality and traceability controls; tooling strategy and maintainability; digital DFM and simulation deliverables; lead-time commitments by gate; risk posture (regional options, business continuity); sustainability (ISCC PLUS readiness, recycled content documentation, carbon accounting practices); and commercial terms (ownership of tooling, maintenance, spares, transfer rights, cost breakdown transparency). Score each criterion with documented evidence.
Capabilities and certifications
Machines, materials, processes
Expect core competencies in thermoplastic injection molding, overmolding, and insert molding, with fluency in runner and gate design, balanced multi-cavity layouts, and scientific molding. For silicone-specific applications, request experience in liquid silicone rubber (LSR) injection molding and compression molding. If you’re exploring silicone parts, you can review process specifics and part types in this overview of custom LSR injection molding parts from JESilicone.
Industry certs and compliance
For medical programs, the U.S. FDA’s Quality Management System Regulation aligns with ISO 13485 and takes effect in February 2026; confirm how a supplier maps procedures to QMSR and any planned audit timelines per the Federal Register final rule and the FDA’s QMSR hub. In automotive, Production Part Approval Process (PPAP) levels and core tools remain foundational; verify experience and submission depth as outlined by AIAG’s Core Tools resources. Aerospace buyers should confirm First Article Inspection competency per IAQG 9102, including familiarity with Forms 1–3 and trigger events.
Quality and traceability
Request documented inspection plans, incoming and lot traceability procedures, calibration and gage R&R practices, and evidence of capability indices at CTQ features during pilots. For regulated programs, specify what you expect at FAI/PPAP/IQ-OQ-PQ gates, including who signs what, and how nonconformances and change control are handled.
A neutral capability example: JESilicone supports custom LSR injection molding, compression molding, and extrusion. For applicable applications, materials can comply with FDA, RoHS, and REACH requirements, and the team has experience with overmolding and plastic injection support. Use vendor documentation to verify specific standards and part-family experience for your program.
Tooling and total cost
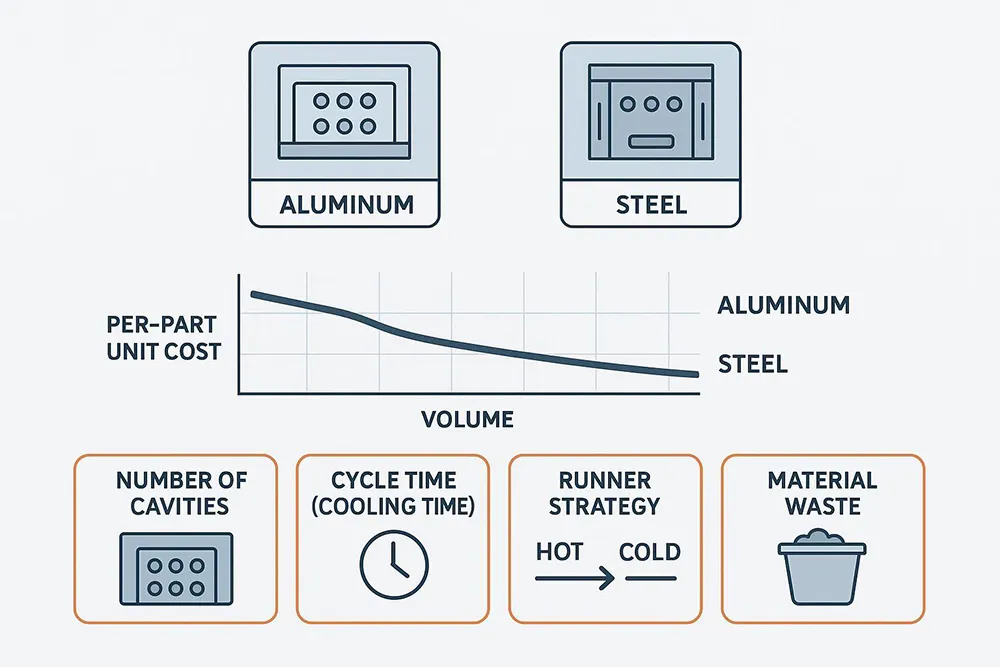
Aluminum vs. steel strategy
Aluminum molds typically offer faster lead times and lower upfront cost—ideal for prototypes, EVT/DVT builds, and early bridge volumes. Steel molds cost more and take longer but deliver longevity (up to millions of cycles), better abrasion resistance for glass-filled resins, and the lowest sustainable unit cost at volume. Practical path? Start in aluminum to learn and stabilize cycle time; then convert to steel for sustained production. Protolabs compares durability, speed, and cost trade-offs across aluminum and steel, while Fictiv adds perspective on material and lifecycle considerations.
Runners, cooling, cycle time
Cycle time is dominated by cooling—often the largest share of the shot. Plastics Technology’s cycle-time analyses show how cooling strategy and gate freeze time drive seconds per part. Conformal cooling and improved circuit design routinely cut cycle time by double digits; Autodesk Moldflow Adviser’s cooling and runner tools help identify baffles, bubblers, and circuit balance opportunities. Hot runners reduce material waste and improve stability in multi-cavity tools, but add maintenance and up-front cost; cold runners are simpler yet can raise material consumption at scale. Model these levers directly in your TCO.
Cost terms and ownership
Clarify in writing: who owns the mold; storage location; preventive maintenance and spares; repair SLAs; and transfer rights. Require a transparent cost breakdown—tooling amortization method, resin and additives, machine-hour rate tied to cycle time, secondary ops, fixture costs, quality/validation charges (FAI/PPAP/IQ-OQ-PQ), logistics/Incoterms, and any warranty/quality allocations. Insist that cycle-time assumptions, scrap rates, and cavity counts are explicit so you can compare quotes apples to apples.
Lead times and validation
Prototypes and bridge tools
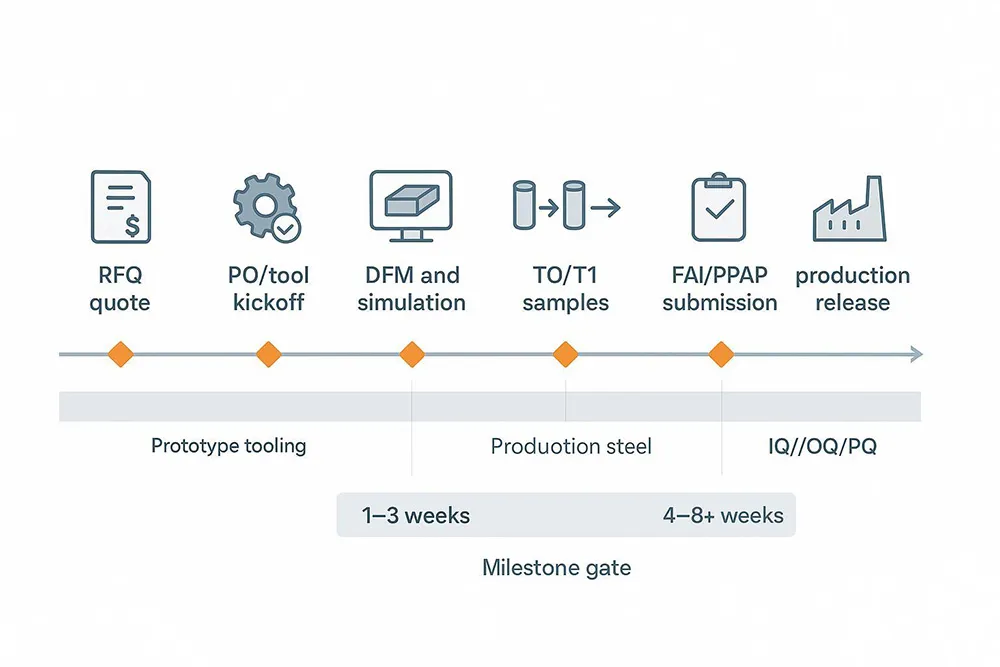
Rapid providers often cite “CAD to parts” in roughly 1–3 weeks for aluminum prototype tools, depending on geometry and finish; production-ready steel molds typically range from about 4–8+ weeks. Treat these as planning bands and validate against your vendors’ current capacity. For context on rapid molding and economic trade-offs, see provider explainers from Fictiv.
FAI, PPAP, IQ/OQ/PQ
In aerospace, plan for a documented First Article Inspection to verify that the part, process, and documentation conform to requirements; IAQG’s public 9102 resources outline the purpose and forms. In automotive, PPAP submission levels (1–5) and 18 core elements govern how you demonstrate production readiness; AIAG’s Core Tools pages define the framework and training. For medical device programs under QMSR/ISO 13485 alignment, structure equipment qualifications and process performance qualification before production release using the FDA’s process validation lifecycle terminology (IQ/OQ/PQ and PPQ concepts). See the IAQG 9102 resource, AIAG Core Tools, and FDA process validation guidance for details.
Digital DFM and simulation
A robust 2026 RFQ asks for digital DFM and simulation outputs: Fill+Pack+Warpage results, gate and runner recommendations, and a cooling analysis summary with estimated cycle time and hotspots. Autodesk’s Moldflow Adviser materials describe runner and cooling analyses you can request. Treat these as negotiation levers: if the model shows a 20–30% cycle-time reduction via cooling improvements or conformal cooling, you can push vendors to reflect that in unit pricing at committed volumes. For capabilities examples, review Autodesk’s Moldflow Adviser brochure.
Sustainability and risk posture
Recycled content and ISCC+
If you plan to claim recycled content via mass balance, confirm site-level ISCC PLUS certification and the supplier’s ability to maintain inputs/outputs/inventory ledgers and issue sustainability declarations. ISCC’s chain-of-custody documents clarify that mass balance must be tracked at the site level and that claims (e.g., percentages) rely on audit-ready records. See ISCC PLUS 203-2 Chain of Custody for specifics.
Carbon accounting readiness
Ask how the supplier compiles Scope 1–3 emissions per the Greenhouse Gas Protocol Corporate Standard and whether they can support product carbon footprints that roll into your corporate inventories. Even a basic energy- and resin-driven estimate improves apples-to-apples comparisons and future reporting.
On/nearshoring resilience
Nearshoring and dual-sourcing can shorten lead times and hedge geopolitical shocks. Deloitte’s manufacturing outlooks describe the sustained momentum behind reshoring and North American supply chains. Weight regional options and transfer-readiness in your rubric so you’re not locked into a single geography. See Deloitte’s manufacturing industry outlook for context.
Shortlisting and RFQ
Scope for apples-to-apples
-
Define EAU and ramp profile; specify resin grade(s), tolerance bands, CTQ features, and cosmetic standards; state gating/parting preferences and any inserts; require dimensional inspection plan, validation deliverables (FAI/PPAP/IQ-OQ-PQ), and a DFM/simulation package; clarify tooling ownership/maintenance/transfer, logistics/Incoterms, sustainability documents (ISCC PLUS declarations; Scope 1–3 summary), and a line-item cost breakdown with cycle-time and scrap assumptions.
Vendor interview checks
-
Probe scientific molding controls, real cooling flow/temperature monitoring, runner balancing approach and gate-freeze rationale; confirm experience with PPAP levels and Cpk targets, FAI/AS9102 forms, and QMSR mapping; ask for recent Moldflow outputs and examples of cycle-time reductions achieved through cooling improvements or conformal cooling.
Scoring and decision gates
-
Weight TCO levers (tooling amortization, cycle time, scrap), validation readiness, lead-time commitments by gate, sustainability maturity, and resilience (regional options/dual-source). Use a documented score to select T0/T1 suppliers, require corrective actions before PPAP/FAI submissions, and set a final release gate contingent on capability indices and run-at-rate performance.
Conclusion
Choosing the right partner in 2026 means modeling TCO explicitly, demanding digital DFM to compress cycle time, confirming validation competence (FAI/PPAP/IQ-OQ-PQ), and testing resilience and sustainability claims with documentation. To move now, standardize your RFQ scope, request simulation outputs with cycle-time assumptions, and schedule supplier interviews against the rubric above. Do that, and you’ll have a defensible, data-backed answer to how to choose an injection molding manufacturer—without surprises on cost, quality, or schedule.