Silicone Tubing for Peristaltic Pumps: Selection, Properties, and Applications

Introduction
Choosing silicone tubing for peristaltic pumps sounds simple—until you realize the tubing is the part that gets squeezed thousands (or millions) of times. The “right” tube helps you keep a steady flow rate, reduce unexpected downtime, and avoid headaches like early cracking or particle shedding.
This guide is for anyone who needs the tubing choice to work in real life: process and design engineers, QA/RA teams who need the paperwork to match the material, and buyers who need consistent supply without constant re-testing.
We’ll walk through a practical, step-by-step way to pick silicone peristaltic pump tubing. We’ll start with sizing and fit (what the pump head can actually handle), then cover material behavior, sterilization and compliance basics, expected service life, and when another tubing material may be a better fit.
Think of it as a selection checklist: confirm the pump head requirements, translate them into ID/OD/wall and hardness, validate with a short trial run, and only then lock the spec for production.
How to choose peristaltic pump tubing - Key essentials
A simple way to choose silicone tubing for peristaltic pumps is to decide in this order:
-
Make sure the tubing fits the pump head.
-
Make sure the tubing can handle your process (temperature, chemicals, pressure, run time).
-
Then worry about paperwork, sterilization, and long-term life.
Fit and tolerances
A peristaltic pump only works as well as the tube it squeezes. If the tubing size is slightly off, you can get flow rate drift, slipping, faster wear, or the tube may fail early.
Start with the pump manufacturer’s recommended tubing sizes. Treat that as your baseline, then confirm your supplier can hold those dimensions consistently.
When you turn a pump requirement into a purchasing spec, capture these items clearly:
-
ID (inner diameter): affects how much fluid moves per revolution.
-
OD and wall thickness: affects how the tube sits in the pump track and how hard the rollers need to squeeze.
-
Ovality and tolerance: “out of round” tubing often shows up as inconsistent flow, especially for dosing.
Grayline highlights how sensitive peristaltic pumps are to wall thickness and fit in its peristaltic pump tubing material selection guide.
What affects flow accuracy
Pump accuracy isn’t only about the controller. It also depends on whether the tube compresses and “springs back” the same way every time.
Key items to specify (and verify with a trial run):
-
Occlusion setting: how fully the rollers pinch the tube closed. Too little can cause slip/backflow; too much shortens tubing life.
-
Hardness (Shore A durometer): softer tubes squeeze easily; harder tubes may handle more backpressure but can need different occlusion settings.
-
Compression set: if the tube stays flattened after being squeezed, your flow rate can drift—especially after the pump sits idle.
Tip: Calibrate the pump using the final tubing you’ll actually use—same lot, same temperature, and after any sterilization step your process requires.
Match the tubing to your process
A tube can look perfect at receiving inspection and still fail once it sees your real conditions. Define these up front:
-
Temperature: normal operation plus any cleaning/sterilization temperatures.
-
Backpressure: restrictions (filters, long lines, valves) make the tubing work harder.
-
Duty cycle: continuous pumping vs. long pauses with the tube held under compression.
-
Fluid + cleaning chemistry: process fluids and cleaning agents can slowly change tubing feel and strength.
-
Particle sensitivity: if particles matter, plan for a realistic wear/particle check.
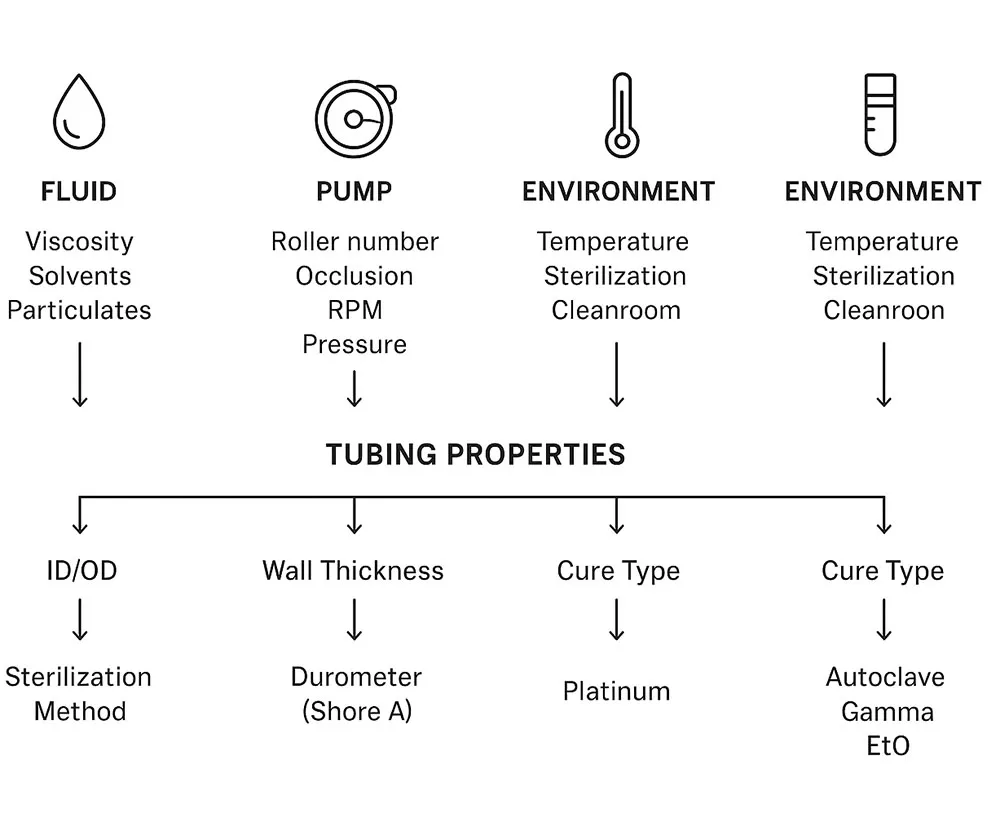
Material Properties of Silicone Peristaltic Pumps Tubing
JESilicone capabilities and quality system
If you need a supplier that can support both prototyping and stable production, it helps to evaluate more than just the tubing spec. JESilicone’s manufacturing and quality setup is designed for regulated and high-consistency use cases:
-
ISO 13485 certified facilities and a 100,000-class clean workshop for medical-grade products.
-
OEM/ODM support: design assistance, prototyping, and mass production for custom tubing and molded parts.
-
Consistent extrusion capability: custom ID/OD, wall thickness, and hardness to match common pump-head requirements.
-
Traceability expectations: lot identification and documentation support (for example, COA and material declarations where applicable).
-
Change control mindset: when you’re qualifying tubing for a validated process, you want advance notice of formulation or process changes.
For product-level starting points and customization options, see JESilicone silicone tubing and custom medical silicone tube.
If you’re sourcing from JESilicone, these pages can help you confirm sizes and customization options: JESilicone silicone tubing and custom medical silicone tube.
This is where silicone tubing for peristaltic pumps stops being a generic material name and becomes a real, testable spec. Two silicone tubes can look similar, but behave very differently once the pump starts squeezing them.
Mechanical behavior that matters
For peristaltic pumping, focus on how the tube handles repeated squeezing:
-
Hardness (Shore A): affects how easily the tube pinches closed and how it handles backpressure.
-
Strength and tear resistance: helps during installation and reduces the chance of small cracks starting under repeated cycles.
-
Compression set: tells you whether the tube “stays flattened” after sitting in the pump head.
-
Dimensional stability: consistent ID/OD/wall helps keep your calibration stable from lot to lot.
If you work in a regulated environment, also think about change control. Even a small formulation tweak can change pump performance over time.
Temperature and chemical reality check
Silicone is known for handling a wide temperature range, which is why it’s popular in labs, food processing, and bioprocessing. Still, it’s best to validate using your actual conditions.
-
Heat (like autoclave/steam): repeated cycles can slowly change flexibility and life.
-
Cold: tubing can feel stiffer, which can affect occlusion and flow rate.
For chemicals, check both the process fluid and what you use for cleaning. Early warning signs of incompatibility include swelling, softening, embrittlement, or a tacky surface.
Gas permeability and particle shedding
Two properties are easy to overlook until they cause problems:
Gas permeability: Silicone lets gases pass through more easily than some specialty tubing. That can matter for oxygen-sensitive fluids or when you need tight gas control.
Spallation (particle shedding): Peristaltic action can shed tiny particles from the tubing surface over long runs. This behavior is discussed in the peer-reviewed study “Spallation of Small Particles From Peristaltic Pump Tube Segments” on PubMed.
Ways to reduce the risk:
-
avoid over-occlusion,
-
keep backpressure as low as practical,
-
replace tubing on a schedule you’ve validated,
-
use filtration when your process allows it (and confirm filter life).
Compliance and sterilization
Important: This article is general guidance only. Always validate tubing performance (flow accuracy, life, particles, and post-sterilization behavior) under your actual pump model, fluid, temperature, backpressure, and duty cycle. For regulated uses, align requirements with your quality system and regulatory strategy—this content isn’t legal, medical, or regulatory advice.
If you use peristaltic pumps in medical, bioprocess, or food applications, compliance isn’t just a label. What you really need is a documentation package that matches your use case.
A practical way to think about it:
-
What it’s made of: material formulation and any declarations of compliance.
-
What it tested like: biocompatibility/extractables (if relevant), and any particulate or performance data you require.
-
What you can trace: lot/COA, change notifications, and traceability expectations.
For food-contact use in the US, teams often reference FDA 21 CFR 177.2600. In purchasing language, it’s usually safer to say “formulated to comply” and “supported by declarations and test data,” rather than “FDA approved.”
For the EU, food-contact compliance is typically handled under Regulation (EC) No 1935/2004 and the GMP Regulation (EC) No 2023/2006. The exact testing and documentation your customer expects can vary by market and application.
Sterilization options
Sterilization is a common place where silicone tubing for peristaltic pumps looks fine on paper but changes after repeat cycles. The goal isn’t just “survive one cycle”—it’s “still performs the same after the number of cycles you plan to use.”
If sterilization is part of your process, define these up front:
-
method (gamma/e-beam, EtO, steam),
-
cycle count,
-
packaging/handling,
-
acceptance checks after sterilization (dimensions + flow calibration + visual inspection).
Common process-level references include:
What to check after sterilization
Common test standards you may see in specs
Depending on your industry and risk assessment, teams may reference common test methods and compendial chapters when writing requirements for silicone tubing for peristaltic pumps:
-
USP <87> and USP <88> are widely used for biological reactivity screening (often discussed as “USP Class VI” testing in supplier documentation).
-
USP <665> and USP <1665> address polymeric components and systems used in pharmaceutical manufacturing.
-
For mechanical properties, suppliers often test elastomers using methods such as ASTM D412 (tensile properties) and ASTM D395 (compression set).
Use these as pointers for what to ask for in a documentation package—your actual requirements should match your application, your customer expectations, and your internal validation plan.
After sterilization, re-check what affects pump performance:
-
ID/OD/wall: even small shifts can change occlusion and delivered volume.
-
Hardness/flexibility: changes can affect calibration and fatigue life.
-
Surface condition: tackiness, haze, microcracks, or deformation can increase wear and particles.
Document your allowed maximum cycle count (based on your validation), and treat that limit as part of the tubing spec.
Service life and optimization
Peristaltic tubing is a consumable. The question isn’t “will it wear out?”—it’s how fast, and how you’ll know it’s time to replace it before it creates quality or downtime issues.
Why service life varies so much
Two systems can use the same tubing and get very different results. The biggest reasons are occlusion setting, backpressure, temperature, and whether the tubing sits in the pump head under compression between runs.
-
Continuous pumping: wear is mostly driven by cycle count and pressure.
-
Intermittent pumping with long idle time: the tube can flatten and recover unevenly, which often shows up as flow drift.
JESilicone supports OEM/ODM tubing extrusion with custom ID/OD, wall thickness, and durometer options—helpful when you want the same fit from prototype runs through production lots.
Common causes of early failure
Most “unexpected” tubing failures are caused by a few repeat issues:
-
Too much occlusion: extra squeeze creates extra heat and fatigue.
-
High backpressure: restrictions make the tube work harder each cycle.
-
High RPM / high cycles: faster cycling accelerates fatigue and particle risk.
-
Hot/cold swings: changes how the tube compresses and rebounds.
-
Chemical mismatch: swelling, softening, embrittlement, or surface damage.
Practical best practices
To get stable results from silicone tubing for peristaltic pumps, aim for repeatability, not “maximum squeeze.”
-
Match the tubing spec to the pump head (size + wall + hardness) and control substitutions through change control.
-
Set occlusion to the minimum that works under your real backpressure.
-
Reduce restrictions where possible (filter sizing, line routing, valves).
-
Choose a replacement rule you can follow (time, cycle count, or drift limit)—not “replace when it breaks.”
-
Include sterilization in validation if you sterilize before use.
Warning: If the formulation, curing system, or extrusion process changes, pump performance can change too. In regulated use, treat supplier changes as a trigger for review or requalification.
Applications and alternatives
Biopharma and medical
In bioprocessing and medical-related fluid handling, silicone is popular because it’s flexible, handles a wide temperature range, and can be supported with the right documentation.
It’s commonly used for:
-
buffer and media transfer at moderate pressures,
-
sampling lines where you want visual inspection,
-
processes where tubing changeout is a normal part of the workflow.
If you’re in a regulated environment, make sure you:
-
define what “acceptable particles” means for your process (if relevant),
-
align traceability and documentation with your quality system,
-
confirm performance after sterilization, not just sterility.
Food and lab
For food processing and labs, silicone tubing is often chosen because it can handle temperature swings and is available in grades designed for repeated food contact. For size and hardness options, JESilicone wholesale silicone rubber tube can be a useful starting point.
When silicone isn’t the best fit
Silicone isn’t always the safest choice if you’re dealing with aggressive solvents, very high pressure, or strict gas-control requirements. In those cases, alternative peristaltic tubing materials may give you longer life or lower process risk.

Use that comparison as a starting point, then confirm with the pump manufacturer’s guidance and a short pilot run that matches your real duty cycle and sterilization plan.
Conclusion
Picking silicone tubing for peristaltic pumps is really about consistency:
-
Fit comes first: the right ID/OD/wall thickness is what keeps flow stable.
-
Then material behavior: hardness, recovery, and compatibility with heat and chemicals.
-
Then compliance and sterilization: the documentation and the post-sterilization checks you actually need.
-
Finally service life: set a replacement rule based on your own validated conditions.
If you want to reduce rework and downtime, lock the tubing spec early, validate it with a short trial run, and keep tight control on substitutions—especially if your process is regulated.